
PDF) Conformational dynamics of the Beta and Kappa SARS-CoV-2 spike proteins and their complexes with ACE2 receptor revealed by cryo-EM
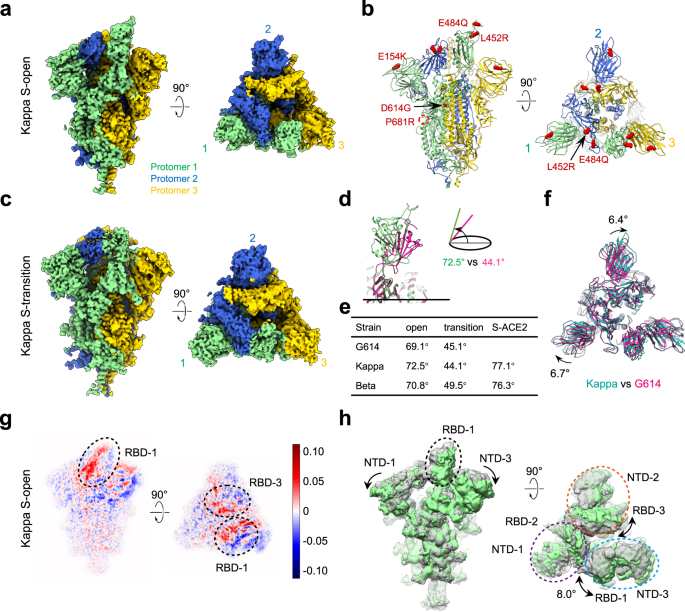
Conformational dynamics of the Beta and Kappa SARS-CoV-2 spike proteins and their complexes with ACE2 receptor revealed by cryo-EM | Nature Communications

Conformational dynamics of the Beta and Kappa SARS-CoV-2 spike proteins and their complexes with ACE2 receptor revealed by cryo-EM. - Abstract - Europe PMC
1 VITA JAMES Z. LEE - LI ZHONGQING PERSONAL Address: Room 3361 Academic Bldg, The Hong Kong University of Science and Technology
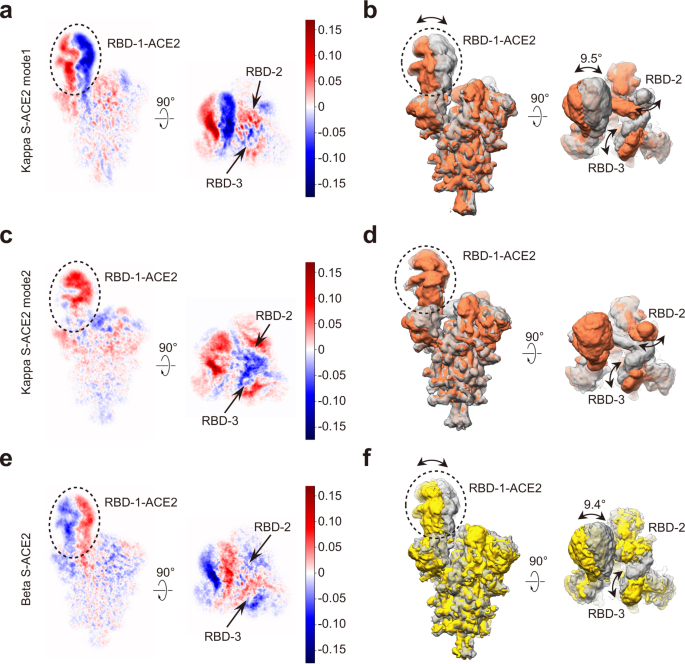
Conformational dynamics of the Beta and Kappa SARS-CoV-2 spike proteins and their complexes with ACE2 receptor revealed by cryo-EM | Nature Communications

Conformational dynamics of the Beta and Kappa SARS-CoV-2 spike proteins and their complexes with ACE2 receptor revealed by cryo-EM | Nature Communications
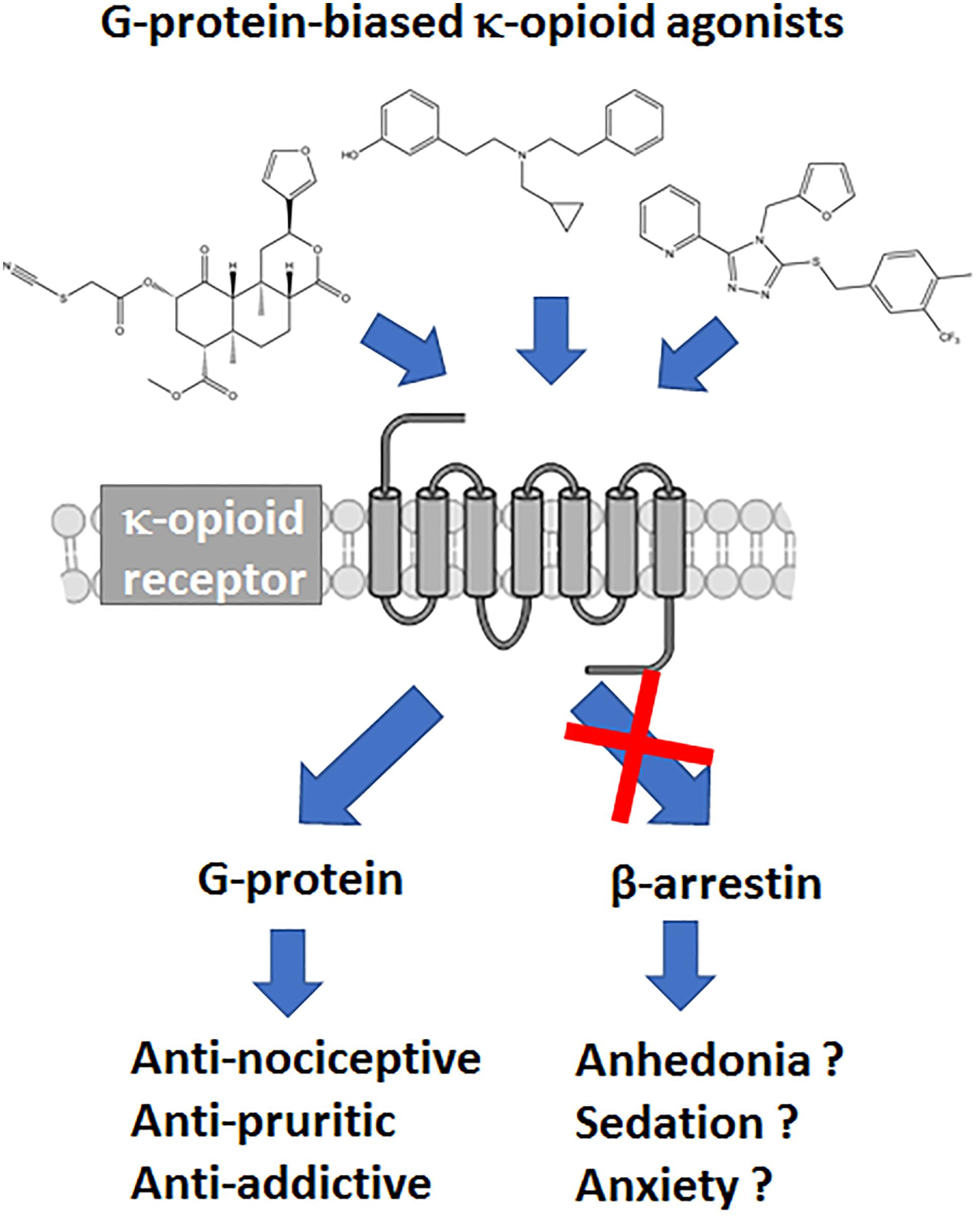
Frontiers | A Review of the Therapeutic Potential of Recently Developed G Protein-Biased Kappa Agonists









